

( Herb medicine for treatment of Diabetic & Diabetic Ulcer )
Julamanee C Plus Capsules
tested in performance and laboratory tests with In-vitro systems and in laboratory animals test to confirm safety Non-toxic to cells And prove the ability to solve problems or treat diabetes and diabetes ulcer with many scientific tests proof
-
Acute Toxicity Tested in Category 5 criteria, thus ensuring safety for consumption
-
Microbial Contamination Test Thus ensuring safety and hygienic good for consumers
-
Inhibition of enzyme activity “Alpha Glucosidase” Which confirms the ability to reduce blood sugar effectively With In-Vitro performance testing system
-
Antibacterial activity test Staphylococcus Aureus & Staphylococcus Epidermidis with In-Vitro Performance Testing System
-
Testing of free radicals With In-Vitro performance testing system
-
Immunity stimulation test With In-Vitro performance testing system
-
The test for stimulation of cell growth in human skin cells With In-Vitro performance testing system
-
Cytotoxicity testing with In-Vitro performance testing system
-
Test of wound healing in human skin cells With In-Vitro performance testing system
-
Matrix Metalloproteinase-2 enzyme inhibition test In human skin cells With In-Vitro performance testing system
Drug characteristics
Is herb powder Through modern production processes and under the control of research and pharmacy teams Packed in a 350 mg capsule containing the size 100 tablets per bottle
How to use
Early Diabetes blood sugar and ulcers:
5 capsules a time, 2 times a day before breakfast and dinner.
Advanced Diabetes blood sugar and ulcers:
5 capsules a time, 4 times a day before breakfast, lunch, dinner and before bedtime.
Warning
Patients with hepatitis or renal failure, pregnant women and children should not take.
Order Now
Why C Plus

Benefits and efficiency Comparison

Suggestion
Take "C Plus" for maximum effectiveness.
Patients should control foods just as body demand per day and should exercise regularly.
Eating more than necessary and lack of exercise will result in decreasing performance, For the good health of all patients.




The table below shows the percentage of accumulated sugar levels and interpretation.
Comparison table for the diagnosis of diabetes with HbA1C (A1C) compared to FBS (mg/dl)





High blood pressure levels

Order Now


Testimonial
 |  |  |
|---|---|---|
 |  |
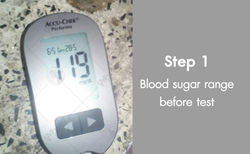
Diabetes and ulcers test subject no.1
Patient Name : Mr.somboon Sriwara
Daily Took 20capsules For 3weeks, Blood Sugar From 119mg/dl Become Lower Than 100mg/dl
 |  |  |
|---|
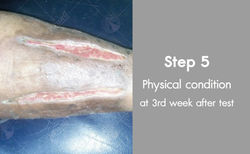
Diabetes and ulcers test subject no.2
Patient Name : Mrs.somjai Ruettidet
Daily Took 10capsules. Before test blood sugar level at 280-350mg/dl After test of 2months blood sugar level at 90-95mg/dl, ulcers healed well
 |  |  |
|---|---|---|
 |